Org. Synth. 1938, 18, 54
DOI: 10.15227/orgsyn.016.0047
METHYL BENZYL KETONE
[2-Propanone, 1-phenyl-]
[(A) (From Phenylacetic and Acetic Acids)]
Submitted by R. M. Herbst and R. H. Manske.
Checked by C. R. Noller and C. F. Love.
1. Procedure
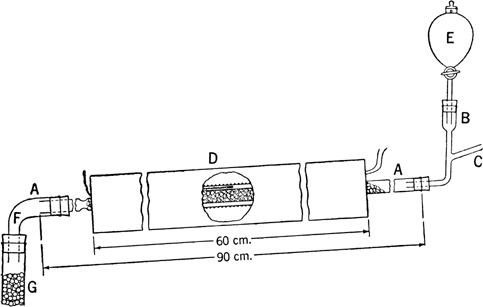
The reaction is carried out in the apparatus shown in
Fig. 13. A-A is a
Pyrex combustion tube, 90 cm. long and 2 cm. in diameter, fitted with an
inlet chamber B, having a sealed-in side arm C and bearing a
separatory funnel E. The center 60 cm. of the tube is filled with thorium oxide catalyst
(Note 1) held in place at the lower end by indentations in the combustion tube. The hot junction D of a
pyrometer is placed in contact with the glass tube at its center, and the catalyst-filled section of the tube is wrapped with a thin layer of asbestos paper. The tube is supported in an
electrically heated cylindrical furnace, 60 cm. in length, which is inclined slightly from the horizontal. The annular space between the tube and the furnace is plugged at the ends with asbestos in order to produce a uniform temperature throughout the tube and to hold the glass tube in place. The lower end of the reaction tube is connected through the
adapter F to a
vertical glass tube G, 40 cm. long and 2 cm. in diameter, which is filled with glass beads and serves as the condenser. An
Erlenmeyer flask or other suitable receiver is placed at the lower end of G.
Fig. 13
The furnace is heated to 430–450°, and simultaneously the tube is swept out thoroughly with a stream of
carbon dioxide, introduced through the side arm C. The
carbon dioxide is passed through a
wash bottle of concentrated sulfuric acid to dry it and to estimate the rate of flow. A solution of
136 g. (1 mole) of phenylacetic acid, m.p.
77–79° (Org. Syn. Coll. Vol. I, 1941, 436), in
120 cc. (120 g., 2 moles) of glacial acetic acid is placed in the separatory funnel E and is introduced into the inlet chamber B at the rate of twelve to fifteen drops per minute. The entire solution should run through in twelve to fifteen hours. Meanwhile a very slow stream of
carbon dioxide (one bubble per second) is passed through the tube C, to keep the gases in motion. After all the solution has been added, the funnel is rinsed with
10 cc. of glacial acetic acid, and this is passed through the reaction tube to facilitate removal of the product. The distillate consists of a slightly fluorescent light brown oil and an aqueous layer; both layers are treated with 300 g. of a mixture of ice and water, and rendered alkaline to litmus with a slight excess of
50 per cent sodium hydroxide solution
(Note 2).
The oil is separated (Note 3), and the aqueous layer is extracted with two 50-cc. portions of benzene. The extracts are combined with the oil, and the solvent is removed by distillation. Fractionation of the residue under reduced pressure gives 80–95 g. of a methyl benzyl ketone fraction, boiling at 110–120°/21–22 mm., and a residue of dibenzyl ketone (Note 4). The main fraction on redistillation yields 74–87 g. (55–65 per cent of the theoretical amount) of methyl benzyl ketone boiling at 110–115°/21–22 mm. (Note 5), (Note 6), and (Note 7).
2. Notes
1.
The catalyst is prepared as follows: Enough pea-sized pieces of screened pumice to fill the tube is soaked in hot concentrated
nitric acid and then washed thoroughly with hot distilled water. In a
porcelain dish the pumice is mixed with a solution of
40 g. of thorium nitrate crystals [Th(NO3)4 + 12H2O] in 100 cc. of water and is evaporated to dryness, with frequent stirring to ensure uniform deposition of the salt. The impregnated pumice is ignited over a
Bunsen burner until decomposition of the nitrate is complete. The pumice carries about
15 g. of thorium oxide.
2.
From the alkaline solution about
10–15 per cent of the phenylacetic acid may be recovered by acidification with
sulfuric acid. The acid separates as an oil which crystallizes slowly on cooling.
3.
Salt may be added to facilitate the separation.
4.
When several runs are made, the residues may be combined and distilled under reduced pressure. The fraction boiling at
190–210°/20 mm. amounts to about
19 g. per run and is chiefly
dibenzyl ketone.
5.
Further purification of the product may be effected by converting the ketone into the bisulfite compound, washing this with
ether, decomposing with
sodium bicarbonate, and steam-distilling.
6.
When several runs are to be made, the catalyst should be regenerated after each run by passing air through the reaction tube for about three hours while the temperature is raised gradually to 550°. The yield in the first run may be low, especially if all the
oxides of nitrogen have not been removed from the reaction tube.
7.
Using the same procedure the following ketones may be obtained in similar yields:
ethyl benzyl ketone from
phenylacetic acid and
propionic acid,
methyl β-phenylethyl ketone from
hydrocinnamic acid and
acetic acid, and
ethyl β-phenylethyl ketone from
hydrocinnamic acid and
propionic acid.
[(B) (From α-Phenylacetoacetonitrile)]
Submitted by Percy L. Julian and John J. Oliver.
Checked by C. R. Noller
1. Procedure
Three hundred fifty cubic centimeters of concentrated sulfuric acid is placed in a
3-l. flask and cooled to −10°. The total first crop of moist
α-phenylacetoacetonitrile obtained according to the procedure on p. 487 (corresponding to 188–206 g., or 1.2–1.3 moles of dry product) is added slowly, with shaking, the temperature being kept below 20°
(Note 1). After all is added the flask is warmed on the
steam bath until solution is complete and then for five minutes longer. The solution is cooled to 0°, 1750 cc. of water added rapidly, and the flask placed on a vigorously boiling
water bath and heated for two hours, with occasional shaking. The ketone forms a layer and, after cooling, is separated and the acid layer extracted with
600 cc. of ether. The oil and ether layers are washed successively with 100 cc. of water, the
ether combined with the oil and dried over
20 g. of anhydrous sodium sulfate. The
sodium sulfate is collected on a filter, washed with
ether, and discarded. The
ether is removed from the filtrates, and the residue distilled from a
modified Claisen flask with a 25-cm. fractionating side arm. The fraction boiling at
109–112°/24 mm. is collected; it weighs
125–150 g. (
77–86 per cent of the theoretical amount)
(Note 2).
2. Notes
1.
If pure dry
α-phenylacetoacetonitrile is used, half its weight of water should be added to the
sulfuric acid or charring will take place on the steam bath.
2.
Usually almost the entire crude product distils in this range with practically no fore-run or residue. Occasionally, however, as much as 30 g. of high-boiling residue, chiefly unchanged nitrile, is obtained. When this happens the yield is correspondingly decreased.
3. Discussion
Methyl benzyl ketone has been prepared by distilling a mixture of the
barium1 or calcium2 salts of phenylacetic and acetic acids; by passing the vapors of these acids over a heated
thorium oxide catalyst;
3,
4 and by heating
phenylacetic acid,
sodium acetate, and
acetic anhydride.
5 Methyl benzyl ketone has also been prepared from
phenylacetyl chloride and
zinc methyl;
6 from
acetyl chloride and
cadmium dibenzyl;
7 by rearrangement of
α-phenyl-β-methylethylene oxide;
8 by heating
α-phenyl-β-methylethylene glycol with dilute
sulfuric acid;
9 by heating the addition product of
chloroacetone and phenylmagnesium bromide;
9 by treating
chloroacetone and benzene with aluminum chloride;
10 and by the hydrolysis of
α-phenylacetoacetic ester11 or phenacylmalonic ester.
12
The procedure in Part (
A) above is based on that of Senderens
3 and Pickard and Kenyon.
4 It has been reported that "thoria aërogel" is superior to other forms of
thoria catalysts for the preparation of ketones from aliphatic acids or esters.
13This preparation is referenced from:
Appendix
Chemical Abstracts Nomenclature (Collective Index Number);
(Registry Number)
oxides of nitrogen
Phenylacetic and Acetic Acids
zinc methyl
cadmium dibenzyl
thoria
sulfuric acid (7664-93-9)
acetic acid (64-19-7)
Benzene (71-43-2)
ether (60-29-7)
acetic anhydride (108-24-7)
sodium acetate (127-09-3)
sodium hydroxide (1310-73-2)
acetyl chloride (75-36-5)
sodium bicarbonate (144-55-8)
nitric acid (7697-37-2)
propionic acid (79-09-4)
sodium sulfate (7757-82-6)
carbon dioxide (124-38-9)
thorium oxide
aluminum chloride (3495-54-3)
Phenylacetic acid (103-82-2)
Phenylmagnesium bromide (100-58-3)
Hydrocinnamic acid (501-52-0)
Methyl benzyl ketone,
2-Propanone, 1-phenyl- (103-79-7)
chloroacetone (78-95-5)
phenylacetyl chloride (103-80-0)
α-phenylacetoacetonitrile (4468-48-8)
dibenzyl ketone (102-04-5)
thorium nitrate (13823-29-5)
ethyl benzyl ketone (1007-32-5)
ethyl β-phenylethyl ketone
α-phenyl-β-methylethylene oxide (14212-54-5)
α-phenyl-β-methylethylene glycol
methyl β-phenylethyl ketone (2550-26-7)
Copyright © 1921-, Organic Syntheses, Inc. All Rights Reserved