Org. Synth. 1972, 52, 33
DOI: 10.15227/orgsyn.052.0033
1,2-DIAROYLCYCLOPROPANES: trans-1,2-DIBENZOYLCYCLOPROPANE
[Methanone, 1,2-cyclopropanediylbis[phenyl]-, trans-]
Submitted by Ismael Colon, Gary W. Griffin, and E. J. O'Connell Jr
1.
Checked by Stephen P. Peters and Kenneth B. Wiberg.
1. Procedure
A 1-l., three-necked, round-bottomed flask equipped with a magnetic stirrer and a dropping funnel is charged with 35 g. (0.14 mole) of 1,3-dibenzoylpropane (Note 1) and a solution of 11.2 g. (0.280 mole) of sodium hydroxide in 400 ml. of methanol. The mixture is warmed to 45°, with stirring to dissolve the diketone. It is allowed to cool to 40°, and a solution of 35 g. (0.14 mole) of iodine in 200 ml. of methanol is slowly added to the stirred solution from the dropping funnel (Note 2). After the addition of iodine is completed, the resulting clear solution is stirred at room temperature for 1.5 hours. During this period, a white solid precipitates (Note 3). The mixture is filtered and the filtrate is placed in a round-bottomed flask. The white solid is washed with four, 100-ml. portions of water and dried at 1 mm. at 57° (1 mm.) for 15 hours, yielding 23–25 g. (66–72%) of trans-1,2-dibenzoylcyclopropane, m.p. 103–104° (Note 4). Additional material is obtained on evaporation of the filtrate using a rotary evaporator. The pale-red solid residue is treated with 25 ml. of aqueous 10% sodium bisulfite (Note 5), filtered, and dried as above giving 8–9 g. (23–26%) of the product, m.p. 95–98°. Recrystallization from methanol affords greater than 90% recovery of trans-1,2-dibenzoylcyclopropane, m.p. 102–103°. For the recrystallization of a 10-g. sample 200 ml. of methanol is used.
2. Notes
2.
The
iodine solution is added at a rate such that the color of the
iodine is continually discharged by the rapidly stirred solution.
3.
In some cases the precipitate may begin to form during the addition of the
iodine solution. This has no effect on the yield.
4.
Pertinent spectral data include a carbonyl stretching band in the IR at 1665 cm.
−1 and UV absorption maxima at 245 nm (ε 31,300), 278 nm (ε 2130), and 317 nm (ε 194). The
1H NMR spectrum (CCl
4) has two triplets of equal intensity at δ 3.32 and 1.68 in addition to the aromatic proton bands at δ 7.3–8.2. The integrated peak areas are in the ratio 1:1:5.
5.
Sodium bisulfite solution is added to reduce unchanged
iodine.
3. Discussion
1,3-Dibenzoylcyclopropane has been prepared by the method described here
2 and previously by Conant and Lutz.
3 Both procedures use
1,3-dibenzoylpropane as the reactant. The present procedure is accomplished in one step under very mild conditions and in nearly quantitative yield. The method of Conant and Lutz is a two-step process involving initial dibromination of the diketone followed by ring closure with
zinc and
sodium iodide, with an overall yield of
trans-1,2-dibenzoylcyclopropane of approximately
15%. The submitters have extended the described method to the preparation of other
trans-1,2-diaroylcyclopropanes, namely
trans-1,2-bis(4-methoxybenzoyl)cyclopropane and
trans-1,2-bis(2-methoxy-5-methylbenzoyl)cyclopropane. In theory the method is applicable to closure of all α,α'-disubstituted propanes having acidic hydrogens on both α-carbons. A probable mechanism for this transformation is as follows:
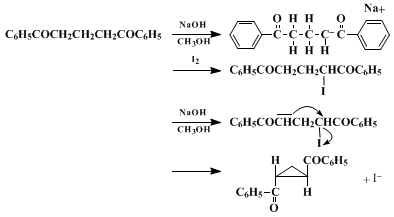
Appendix
Chemical Abstracts Nomenclature (Collective Index Number);
(Registry Number)
methanol (67-56-1)
sodium hydroxide (1310-73-2)
sodium bisulfite (7631-90-5)
iodine (7553-56-2)
zinc (7440-66-6)
sodium iodide (7681-82-5)
1,4-Dibenzoylbutane (3375-38-0)
1,3-dibenzoylpropane (6263-83-8)
1,3-Dibenzoylcyclopropane
trans-1,2-Dibenzoylcyclopropane,
Methanone, 1,2-cyclopropanediylbis[phenyl]-, trans- (38400-84-9)
trans-1,2-bis(4-methoxybenzoyl)cyclopropane
trans-1,2-bis(2-methoxy-5-methylbenzoyl)cyclopropane
Copyright © 1921-, Organic Syntheses, Inc. All Rights Reserved