Org. Synth. 1997, 74, 147
DOI: 10.15227/orgsyn.074.0147
[3+2]-ANIONIC ELECTROCYCLIZATION USING 2,3-BIS(PHENYLSULFONYL)-1,3-BUTADIENE: trans-4,7,7-TRICARBOMETHOXY-2-PHENYLSULFONYLBICYCLO[3.3.0]OCT-1-ENE
[Benzene, 1,1'-[[1,2-bis(methylene)-1,2-ethanediyl]bis(sulfonyl)]bis-]
Submitted by Albert Padwa
1, Scott H. Watterson, and Zhijie Ni.
Checked by David Young, Michael Rupp, Dan Kuzmich, and David J. Hart.
1. Procedure
A. 2,3-Bis(phenylsulfinyl)-1,3-butadiene. In a 2-L, three-necked, round-bottomed flask equipped with a mechanical stirrer, 25-mL dropping funnel, and a nitrogen inlet, are placed 7.75 g (0.09 mol) of 2-butyne-1,4-diol (Note 1), 37.6 mL (0.27 mol) of triethylamine, and 700 mL of dichloromethane. After the diol is completely dissolved, the mixture is cooled to −78°C. To this solution is added 26.0 g (0.18 mol) of phenylsulfenyl chloride (Note 2) over a 30-min interval. The mixture is gradually warmed to room temperature and stirred for an additional 12 hr. The solution is then washed with 100 mL each of water, saturated ammonium chloride solution, saturated sodium bicarbonate solution, and brine, and dried over sodium sulfate. After filtration and concentration, the residue is recrystallized from methanol-ether (1:1) to give 20.1 g (74%) of 2,3-bis(phenylsulfinyl)-1,3-butadiene as a 1.1:1 mixture of diastereomers (Note 3).
B.
2,3-Bis(phenylsulfonyl)-1,3-butadiene (1). A solution of
17.0 g of 2,3-bis(phenylsulfinyl)-1,3-butadiene (56.3 mmol) and
34 mL of hydrogen peroxide (30–35%) (Note 4) in
170 mL of glacial acetic acid is placed in a
500-mL, three-necked flask equipped with a
reflux condenser and
thermometer. The solution is warmed using an
oil bath such that a reaction temperature of 90°C is maintained for 5 hr
(Note 5). To the hot solution is added 40 mL of water and the mixture is allowed to stand at room temperature for 12 hr. The resulting precipitate is collected, washed with water (50 mL), cold
methanol (30 mL), and
ether (100 mL). The filter cake (13.9 g) is dissolved in
100 mL of hot dichloromethane and
100 mL of hexane is added slowly. After standing at 25°C for 1 day, the crystalline solid is collected and washed with
100 mL of ether to give
11.6 g (
61%) of
2,3-bis(phenylsulfonyl)-1,3-butadiene (Note 6).
2 Concentration and recrystallization of the mother liquor affords an additional
1.7 g (
9%) of the disulfone.
C. Dimethyl (E)-5-methoxycarbonyl-2-hexenedioate. Into a flame-dried, 500-mL, three-necked, round-bottomed flask equipped with a magnetic stirring bar, a 100-mL dropping funnel, and a nitrogen inlet, is placed 4.2 g of sodium hydride (60% in oil, 105 mmol) which has been washed twice with 50 mL of hexane and then suspended in 400 mL of anhydrous tetrahydrofuran (THF). The mixture is cooled to 0°C with an ice bath and a solution containing 13.7 mL (15.8 g, 120 mmol) of dimethyl malonate in 30 mL of anhydrous THF is added over a 30-min interval. After being stirred for an additional 20 min at 0°C, the mixture is transferred by cannula into an ice-cold solution containing 19.9 g (110 mmol) of methyl 4-bromocrotonate (Note 7) in 300 mL of THF in a 1-L, round-bottomed flask fitted with a nitrogen inlet. Stirring is continued for an additional 20 min and the reaction is quenched by the addition of 100 mL of a saturated ammonium chloride solution. The solvent is removed with a rotary evaporator at aspirator vacuum, and the aqueous layer is extracted three times with ether (200 mL). The organic layer is washed with 100 mL each of water and brine, and dried over sodium sulfate. After filtration and concentration, the resulting residue is distilled using a 5-cm Vigreux column under reduced pressure to give 8.8 g (38%) of dimethyl (E)-5-methoxycarbonyl-2-hexenedioate (Note 8).
D. trans-4,7,7-Tricarbomethoxy-2-phenylsulfonylbicyclo[3.3.0]oct-1-ene. In a flame-dried, 2-L, three-necked, round-bottomed flask equipped with a magnetic stirrer, dropping funnel, and nitrogen inlet, is placed 1.44 g of sodium hydride (60% in oil, 36.0 mmol) which has been washed twice with 50 mL of hexane and then suspended in 500 mL of anhydrous THF. After 7.59 g (33.0 mmol) of dimethyl (E)-5-methoxycarbonyl-2-hexenedioate in 50 mL of THF is added to the above mixture, it is stirred at 0°C for 30 min. Then a solution containing 10.02 g (30.0 mmol) of 2,3-bis(phenylsulfonyl)-1,3-butadiene in 900 mL of anhydrous THF is added over 30 min. The solution is stirred for 10 min at 0°C and then quenched with 100 mL of a saturated aqueous ammonium chloride solution. The solvent is removed under reduced pressure, and the residue is extracted three times with 300 mL of dichloromethane. The organic phase is washed with 100 mL each of water, brine, and dried over sodium sulfate. The filtrate is concentrated under reduced pressure and the residue is chromatographed on 350 g of silica gel eluting with 4 L of a 2:1-hexane/ethyl acetate mixture to give 9.54 g (75%) of trans-4,7,7-tricarbomethoxy-2-phenylsulfonylbicyclo[3.3.0]oct-1-ene as a clear oil (Note 9).
2. Notes
1.
2-Butyne-1,4-diol, sodium hydride (60% in oil) and dimethyl malonate were purchased from Aldrich Chemical Company, Inc. and used without further purification.
Triethylamine and
dichloromethane were distilled from
calcium hydride prior to use.
2.
Phenylsulfenyl chloride was prepared according to the known literature procedure.
33.
This product is a 1.1:1 mixture of two diastereoisomers, mp
127–129°C, and was used in the next step without further purification. The product has the following spectral properties:
1H NMR (200 MHz, CDCl
3) δ: 5.6, 5.85, 6.18, 6.22 (four s, 4 H), 7.1–7.5 (m, 10 H).
4.
Hydrogen peroxide (30–35%) and glacial acetic acid were purchased from Fisher Scientific Company and were used without further purification.
5.
The yield is considerably reduced if the reaction temperature is allowed to exceed 90°C. Yields as high as
80% have been obtained with careful monitoring of the temperature.
6.
The product has the following physical properties: mp
181–183°C (lit.
2 183–185°C); IR (KBr) cm
−1: 3120, 1580, 1441, 1302, 1140, 1067, 744, 685;
1H NMR (300 MHz, CDCl
3) d: 6.61 (s, 2 H), 6.74 (s, 2 H), 7.20–7.30 (m, 4 H), 7.42–7.51 (m, 6 H);
13C NMR (75 MHz, CDCl
3) δ: 128.4 (d), 129.1 (d), 130.8 (t), 133.7 (d), 137.1 (s), 140.8 (s). Anal. Calcd for C
16H
14O
4S
2: C, 57.46; H, 4.22; S, 19.18. Found: C, 57.39; H, 4.23; S, 19.08.
7.
Methyl 4-bromocrotonate was purchased from Lancaster Synthesis Inc. and distilled before use.
8.
Dimethyl (E)-5-methoxycarbonyl-2-hexenedioate4 has the following physical properties: bp
144–146°C (2.0 mm); IR (neat) cm
−1: 3004, 2947, 1737, 1431, 1268, 1033, 727;
1H NMR (300 MHz, CDCl
3) δ: 2.79 (t, 2 H, J = 7.2), 3.51 (t, 1 H, J = 7.2), 3.70 (s, 3 H), 3.74 (s, 6 H), 5.98 (d, 1 H, J = 15.6), 6.86 (dt, 1 H, J = 15.6, 7.2);
13C NMR (75 MHz, CDCl
3) δ: 30.9, 50.1, 51.3, 52.6, 123.3, 143.7, 166.1, 168.5. This compound has also been prepared by the ozonolysis of
dimethyl allylmalonate followed by reaction with
methyl (triphenylphosphoranylidene)acetate.
5 While this procedure is reported to afford the triester in
60–65% overall yield, we found the above method to be simpler and more convenient for the preparation of larger quantities of material.
9.
trans-4,7,7-Tricarbomethoxy-2-(phenylsulfonyl)bicyclo[3.3.0]oct-1-ene has the following spectral properties: IR (neat) cm
−1: 3004, 2947, 1730, 1438, 1147, 1075, 727;
1H NMR (300 MHz, CDCl
3) δ: 1.86 (t, 1 H, J = 12.3), 2.56 (dd, 1 H, J = 13.2, 7.8), 2.79–2.99 (m, 3 H), 3.17 (brd, 1 H, J = 21.0), 3.34 (brd, 1 H, J = 21.0), 3.4–3.7 (m, 1 H), 3.56 (s, 3 H), 3.65 (s, 3 H), 3.67 (s, 3 H), 7.47 (t, 2 H, J = 7.5), 7.56 (t, 1 H, J = 7.2), 7.78 (d, 2 H, J = 7.5);
13C NMR (75 MHz, CDCl
3) δ: 33.2, 37.4, 39.4, 48.9, 51.9, 52.8, 53.0, 54.7, 63.1, 127.3, 129.0, 129.1, 133.4, 139.7, 161.0, 170.7, 171.2, 172.4; HRMS Calcd for C
20H
22O
8S: m/e 422.1035. Found m/e 422.1030.
Handling and Disposal of Hazardous Chemicals
The procedures in this article are intended for use only by persons with prior training in experimental organic chemistry. All hazardous materials should be handled using the standard procedures for work with chemicals described in references such as "Prudent Practices in the Laboratory" (The National Academies Press, Washington, D.C., 2011 www.nap.edu). All chemical waste should be disposed of in accordance with local regulations. For general guidelines for the management of chemical waste, see Chapter 8 of Prudent Practices.
These procedures must be conducted at one's own risk. Organic Syntheses, Inc., its Editors, and its Board of Directors do not warrant or guarantee the safety of individuals using these procedures and hereby disclaim any liability for any injuries or damages claimed to have resulted from or related in any way to the procedures herein.
3. Discussion
The chemistry of phenylsulfonyl-substituted 1,3-butadienes is receiving increasing attention because of their synthetic versatility and the efficient π-bond activation by the sulfonyl group.
6,7,8,9,10 Some of the procedures currently available for their preparation include phenylsulfonyl-mercuration of 1,3-dienes,
11 condensation of allyl sulfones with aldehydes followed by acetylation and subsequent elimination,
12 thermal SO
2 extrusion from 2-(arylsulfonyl)sulfolenes,
13,14 cheletropic ring-opening of sulfolanes,
15 palladium(II)-catalyzed chloroacetoxylation of 1,3-dienes,
16 and the
2-tosylvinyl sulfone coupling with vinylstannanes.
17 Methods of preparing bis(phenylsulfonyl)-substituted 1,3-dienes, however, are limited to relatively few routes.
18 In spite of its simplicity and its obvious potential as an activated diene,
2,3-bis(phenylsulfonyl)-1,3-butadiene (1) has not been used extensively in organic synthesis. This reagent was prepared by a modification of the procedure of Jeganathan and Okamura in multigram quantities.
2 Treatment of
2-butyne-1,4-diol with
benzenesulfenyl chloride produces the disulfenate ester as a transient species, which rapidly undergoes a series of 2,3-sigmatropic rearrangements to give
2,3-bis(phenylsulfinyl)-1,3-butadiene. This material is readily oxidized with
hydrogen peroxide to the bis(phenylsulfonyl)diene
1 in
70–80% yield.
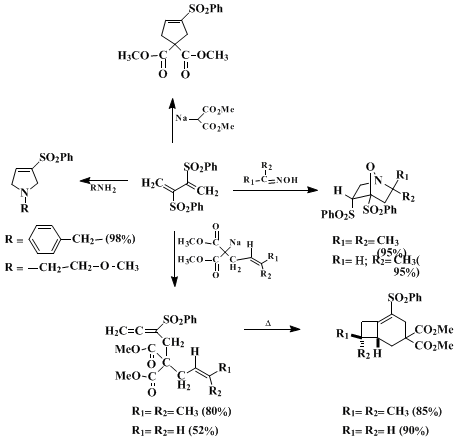
Over the past several years, the submitter has demonstrated the use of diene
1 as a versatile building block in organic synthesis, particularly for [4+2]-cycloaddition chemistry.
19 In an early report, the cycloaddition of this diene with various oximes as a method for
piperidine formation was described.
20 In subsequent investigations, disulfone
1 was demonstrated to undergo cycloaddition to a variety of simple imines
21 and enamines
22 under mild conditions producing novel rearranged heterocycles. This diene also played an important role in the successful outcome of the submitter's [4+1]-annulation strategy for
pyrrolidine formation, since it is highly activated toward nucleophilic addition.
23 More recently, disulfone
1 was found to undergo a [4+1]-annulation reaction with a variety of soft carbanions to give phenylsulfonyl-substituted cyclopentenes in good yield.
24 The pivotal step in this reaction involves addition of a stabilized carbanion onto the highly activated π-bond of diene
1 followed by PhSO
2-elimination to give a phenylsulfonyl-substituted allene as a key intermediate. Further reaction of the allene with benzenesulfinate anion eventually provides the five-membered ring. Several phenylsulfonyl alkenyl-substituted allenes were also prepared and found to undergo a highly chemo- and stereospecific intramolecular [2+2]-cycloaddition reaction to give bicyclo[4.2.0]oct-1-enes in high yield.
25The procedure described here provides a simple and general approach for the construction of bicyclo[3.3.0]oct-1-enes using
2,3-bis(phenylsulfonyl)-1,3-butadiene (1) as a key reagent. The [3+2]-cycloaddition of an allyl anion with an activated olefin to give cyclopentyl anion has been an active area of investigation for many years.
26 The synthetic potential of disulfone
1 for [3+2]-cyclization chemistry is demonstrated here by taking advantage of two properties of the molecule: (1) the ability of the phenylsulfonyl group to activate the double bond toward Michael addition with soft β-dicarbonyl anions and (2) the facility with which the phenylsulfonyl group can be eliminated. Thus, conjugate addition of the anion derived from
dimethyl (E)-5-methoxycarbonyl-2-hexenedioate to diene
1 is followed by a series of consecutive tandem Michael additions to the activated π-bonds.
27 Eventual phenylsulfinate elimination produces the bicyclo[3.3.0]oct-1-ene skeleton. Since disulfone
1 can react with a variety of β-dicarbonyl anions, a wide assortment of bicyclo[3.3.0]octenes is easily available. This strategy can clearly be applied to more complex targets.
Appendix
Chemical Abstracts Nomenclature (Collective Index Number);
(Registry Number)
silica gel
brine
acetic acid (64-19-7)
ethyl acetate (141-78-6)
methanol (67-56-1)
ether (60-29-7)
ammonium chloride (12125-02-9)
sodium bicarbonate (144-55-8)
sodium sulfate (7757-82-6)
piperidine (110-89-4)
hydrogen peroxide (7722-84-1)
dichloromethane (75-09-2)
Tetrahydrofuran,
THF (109-99-9)
pyrrolidine (123-75-1)
sodium hydride (7646-69-7)
hexane (110-54-3)
triethylamine (121-44-8)
calcium hydride (7789-78-8)
Benzenesulfenyl chloride,
Phenylsulfenyl chloride (931-59-9)
Palladium(II)
dimethyl malonate (108-59-8)
methyl 4-bromocrotonate (1117-71-1)
methyl (triphenylphosphoranylidene)acetate (2605-67-6)
2,3-Bis(phenylsulfonyl)-1,3-butadiene,
Benzene, 1,1'-[[1,2-bis(methylene)-1,2-ethanediyl]bis(sulfonyl)]bis- (85540-20-1)
trans-4,7,7-Tricarbomethoxy-2-phenylsulfonylbicyclo[3.3.0]oct-1-ene,
trans-4,7,7-Tricarbomethoxy-2-(phenylsulfonyl)bicyclo[3.3.0]oct-1-ene
2,3-Bis(phenylsulfinyl)-1,3-butadiene (85540-18-7)
Dimethyl (E)-5-methoxycarbonyl-2-hexenedioate (93279-60-8)
dimethyl allylmalonate (40637-56-7)
2-tosylvinyl sulfone
2-butyne-1,4-diol (110-65-6)
Copyright © 1921-, Organic Syntheses, Inc. All Rights Reserved