Org. Synth. 2000, 77, 153
DOI: 10.15227/orgsyn.077.0153
9-ETHYL-3,6-DIMETHYLCARBAZOLE (DMECZ)
[
9H-Carbazole, 9-ethyl-3,6-dimethyl-
]
Submitted by Jason R. Buck, Minnie Park, Zhiwei Wang, Daniel R. Prudhomme, and Carmelo J. Rizzo
1
.
Checked by Gilles Chambournier and David J. Hart.
1. Procedure
A.
3,6-Dibromo-9-ethylcarbazole
. In an
oven dried, 1-L, round-bottomed flask equipped with a
magnetic stir bar and a
rubber septum are placed
3,6-dibromocarbazole (10.0 g, 31.0 mmol)
(Note 1) and
sodium hydroxide pellets (1.2 g, 30.0 mmol) in
500 mL of dry acetone
(Note 2) under an
argon atmosphere.
Diethyl sulfate (4.1 mL, 31.0 mmol)
(Note 3) is added dropwise over 15 min to the stirred reaction mixture at room temperature. After the addition, the reaction is stirred for 8 hr at which time all solids are removed by filtration and the solvent is removed under reduced pressure to give a yellow solid. The residue is dissolved in
ethyl acetate (200 mL) and successively washed with aqueous saturated
sodium bicarbonate (3 × 75 mL),
brine (3 × 75 mL) and water (3 × 75 mL). The organic layer is dried over
sodium sulfate
, filtered and the solvent removed under reduced pressure to give a pale yellow solid. Recrystallization from
95%
ethanol (ca. 350 mL) gives
3,6-dibromo-9-ethylcarbazole
(
8.85 g,
81% yield) as white needles, mp
139-141°C (lit.
2 mp
137-138°C)
(Note 4). The filtrate is evaporated and the resulting solid recrystallized to give additional product (
0.24 g,
2% yield).
B.
9-Ethyl-3,6-dimethylcarbazole
. In an oven-dried, 1-L, three-necked, round-bottomed flask equipped with a magnetic stir bar, a
reflux condenser and a rubber septum are placed
3,6-dibromo-9-ethylcarbazole (7.1 g, 20.0 mmol) and
[1,3-bis(diphenylphosphino)propane]nickel(II) chloride (0.54 g, 1.0 mmol)
(Note 5) in
500 mL of dry ether
(Note 6) under an
argon atmosphere
(Note 7). To the stirred solution at room temperature is added
methylmagnesium bromide (30 mL, 60.0 mmol)
(Note 8) dropwise over 20 min via an
addition funnel. During the addition, the color of the solution turns from orange to yellow to brown. After the addition, the reaction mixture is heated at reflux for 2 hr at which time the reaction is judged complete by TLC analysis. The reaction mixture is cooled to room temperature, then carefully quenched with aqueous saturated
ammonium chloride (25 mL) at which point a brown precipitate forms. All the contents of the reaction are transferred to a
separatory funnel and successively washed with aqueous saturated
sodium bicarbonate (3 × 50 mL),
brine (3 × 50 mL) and deionized water (3 × 50 mL). The combined aqueous layers are extracted with
ethyl acetate (3 × 50 mL). The combined organic extracts are dried over
sodium sulfate, filtered and the solvent is removed under reduced pressure to give a yellow solid. Recrystallization from
ethanol gives
9-ethyl-3,6-dimethylcarbazole
(
3.62 g,
81% yield) as white needles, mp
57-58°C (lit.
2 mp
62-63°C)
(Note 9). The filtrate is evaporated and the resulting solid recrystallized from
ethanol to give additional product (
0.39 g,
9% yield).
2. Notes
1.
3,6-Dibromocarbazole (99%) was obtained from Aldrich Chemical Company, Inc.
, and was recrystallized from
ethanol before use.
2.
ACS grade
acetone
was dried over activated four angstrom molecular sieves.
3.
Diethyl sulfate (HIGHLY TOXIC; CANCER SUSPECT AGENT) was obtained from Aldrich Chemical Company, Inc.
, and used as received.
4.
The spectra are as follows:
1H NMR (CDCl
3) δ: 1.39 (t, 3 H, J = 7.2), 4.29 (q, 2 H, J = 7.2), 7.26 (d, 2 H, J = 8.7), 7.55 (dd, 2 H, J = 8.7, 1.9), 8.12 (d, 2 H, J = 1.9)
;
13C NMR (CDCl
3) δ: 13.7 (CH
3), 37.8 (CH
2), 110.1 (CH), 111.9 (C), 123.3 (CH), 123.5 (C), 129.0 (CH), 138.8 (C)
; MS (EI): m/e (relative intensity) 355 (48), 353 (100), 351 (52), 340 (48), 338 (92), 336 (46)
. Anal. Calcd for C
14H
11Br
2N: C, 47.84; H, 3.16. Found: C, 47.63; H, 3.13.
5.
[1,3-Bis(diphenylphosphino)propane]nickel(II) chloride (99%) was purchased from Strem Chemical
and used as received.
6.
Diethyl ether was freshly distilled from a
sodium/
benzophenone ketyl.
7.
Gentle warming with a heat gun may be required to dissolve completely
3,6-dibromo-9-ethylcarbazole in
ether.
8.
Methylmagnesium bromide was purchased from Aldrich Chemical Company, Inc.
as a 3.0 M solution in
diethyl ether and used as received.
9.
The spectra are as follows:
1H NMR (CDCl
3) δ: 1.14 (t, 3 H, J = 7.2), 2.58 (s, 6 H), 4.34 (q, 2 H, J = 7.2), 7.31 (s, 4 H), 7.91 (s, 2 H)
;
13C NMR (CDCl
3) δ: 13.7 (CH
3), 21.3 (CH
3), 37.5 (CH
2), 108.0 (CH), 120.3 (CH), 122.8 (C), 126.7 (CH), 127.6 (C), 138.4 (C)
; MS (EI): m/e (relative intensity) 223 (69), 208 (100)
. Anal. Calcd for C
16H
17N: C, 86.05; H, 7.68. Found: C, 85.35; H, 7.70.
Handling and Disposal of Hazardous Chemicals
The procedures in this article are intended for use only by persons with prior training in experimental organic chemistry. All hazardous materials should be handled using the standard procedures for work with chemicals described in references such as "Prudent Practices in the Laboratory" (The National Academies Press, Washington, D.C., 2011 www.nap.edu). All chemical waste should be disposed of in accordance with local regulations. For general guidelines for the management of chemical waste, see Chapter 8 of Prudent Practices.
These procedures must be conducted at one's own risk. Organic Syntheses, Inc., its Editors, and its Board of Directors do not warrant or guarantee the safety of individuals using these procedures and hereby disclaim any liability for any injuries or damages claimed to have resulted from or related in any way to the procedures herein.
3. Discussion
Since its introduction by Barton and McCombie, the deoxygenation of thionocarbonyl derivatives of alcohols has become an important synthetic reaction and a valuable method for the generation of carbon-centered radicals.
3,4
5 Xanthates, thionobenzotes, thionocarbonyl imidazolides, aryloxy thionocarbonate, N-phenylthionocarbamates and oxalate esters are conveniently deoxygenated with tin or silicon hydrides in boiling
benzene or
toluene.
4,5,6
7
8
9
10
11
Saito demonstrated that benzoates and m-(trifluoromethyl)benzoates of secondary alcohols could be deoxygenated via a photoinduced electron-transfer (PET) mechanism using
9-methylcarbazole (MCZ) as the electron donor.
12,13
14 The mechanism involves an excited state electron-transfer from MCZ to the benzoate to give the MCZ/benzoate radical ion pair; solvent cage escape is promoted by salts such as
magnesium perchlorate. Protonation of the benzoate radical anion gives radical
1, which undergoes β-scission to the deoxygenation radical. Hydrogen atom transfer from the solvent (i.e.,
2-propanol) gives the deoxygenated product. Oxidation of the solvent radical by MCZ radical cation regenerates the donor. In principle, the donor could be used in substoichiometric amounts since it is regenerated; however, in practice one full equivalent of MCZ is necessary for the PET deoxygenation.
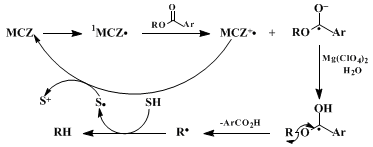
The submitters reasoned that the radical cation of MCZ was undergoing side reactions or degradation faster than the steps leading to the regeneration of MCZ. The radical cations of carbazoles have been previously studied by cyclic voltammetry (CV) and it was found that MCZ was irreversibly oxidized, indicating that the radical cation undergoes side reactions faster than the CV time scale.
2
15 The electrochemical oxidation of some 9-alkyl carbazoles substituted at the 3- and 6-positions showed improved reversibility, indicating that the radical cations were longer-lived. The submitters demonstrated that
10-20 mol % of 9-ethyl-3,6-dimethylcarbazole (DMECZ) could efficiently deoxygenate benzoate and m-(trifluoromethyl)benzoates in high yields (Table).
16 The increased lifetime of the radical cation of DMECZ allows for the donor to be regenerated and thus it can be used in substoichiometric amounts. Importantly, it appears that DMECZ is a more reactive donor. Because of the toxicity of tin species, there has been interest in developing alternative methods for the Barton and related deoxygenation reactions and some success has been achieved.
17
18
19
20 The photodeoxygenation not only avoids toxic tin species, but also it is conducted in relatively benign solvents (
2-propanol/water). In addition, the deoxygenation is carried out at room temperature or below, and benzoyl derivatives are attractive because of their easy synthetic access under mild and neutral conditions.
TABLE
PHOTOINDUCED ELECTRON-TRANSFER DEOXYGENATION OF BENZOATES AND m-(TRIFLUOROMETHYL)BENZOATES WITH 9-ETHYL-3,6-DIMETHYLCARBAZOLE
|
|
Substrate
|
Product and Yield
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
9-Ethyl-3,6-dimethylcarbazole was previously synthesized in four steps beginning with the formylation of
9-ethylcarbazole
to give
9-ethylcarbazole-3-carboxaldehyde which is now commercially available.
21 Wolff-Kishner reduction, formylation of the 6-position, and a second Wolff-Kishner reduction gives DMECZ. The reported overall yield for the final three steps is 45%. The present procedure provides quantities of the desired compound in high overall yield by a shorter, more convenient sequence. The desired material was also prepared by a nickel-catalyzed cross-coupling of
3,6-dibromo-9-ethylcarbazole with
methylmagnesium bromide (Corriu-Kumada coupling).
22
23
3,6-Dibromo-9-ethylcarbazole was prepared by N-alkylation of commercially available
3,6-dibromocarbazole with
diethyl sulfate. Alternatively,
3,6-dibromo-9-ethylcarbazole could be prepared by the bromination of
9-ethylcarbazole (Br
2,
acetic acid, 0°C); however, this method also produced significant quantities of
1,3,6-tribromo-9-ethylcarbazole.
Acknowledgments: This work was supported by Grant #DHP-172 from the American Cancer Society.
This preparation is referenced from:
Appendix
Chemical Abstracts Nomenclature (Collective Index Number);
(Registry Number)
9-Ethyl-3,6-dimethyl-carbazole:
9H-Carbazole, 9-ethyl-3,6-dimethyl- (9); (51545-42-7)
3,6-Dibromo-9-ethylcarbazole:
Carbazole, 3,6-dibromo-9-ethyl- (8);
9H-Carbazole, 3,6-dibromo-9-ethyl- (9); (33255-13-9)
3,6-Dibromocarbazole:
Carbazole, 3,6-dibromo- (8);
9H-Carbazole, 3,6-dibromo- (9); (6825-20-3)
Diethyl sulfate: HIGHLY TOXIC; CANCER SUSPECT AGENT:
Sulfuric acid, diethyl ester (8,9);
(64-67-5)
[1,3-Bis(diphenylphosphino)propane]nickel(II) chloride: CANCER SUSPECT AGENT:
Nickel, dichloro[trimethylenebis[diphenylphosphine]]- (8);
Nickel, dichloro[1,3-propanediylbis[diphenylphosphine]-P,P'}- (9); (15629-92-2)
Methylmagnesium bromide:
Magnesium, bromomethyl- (8,9); (75-16-1)
Copyright © 1921-, Organic Syntheses, Inc. All Rights Reserved